 
The emitted frequency of radiation is determined by the energy differences in the atom. So an orbiting electron should radiate energy continuously, causing the electron to spiral into the nucleus ( Figure 3).īohr broke with classical physics by stating that the electron doesn’t radiate light while it accelerates around the nucleus radiation of light occurs only when the electron makes a transition from a higher energy level to a lower energy level. An electron that orbits a nucleus is constantly accelerating. Accelerated electrons, according to James Clerk Maxwell’s theory, radiate energy in the form of electromagnetic waves. The classical view of the atom had a glaring difficulty. 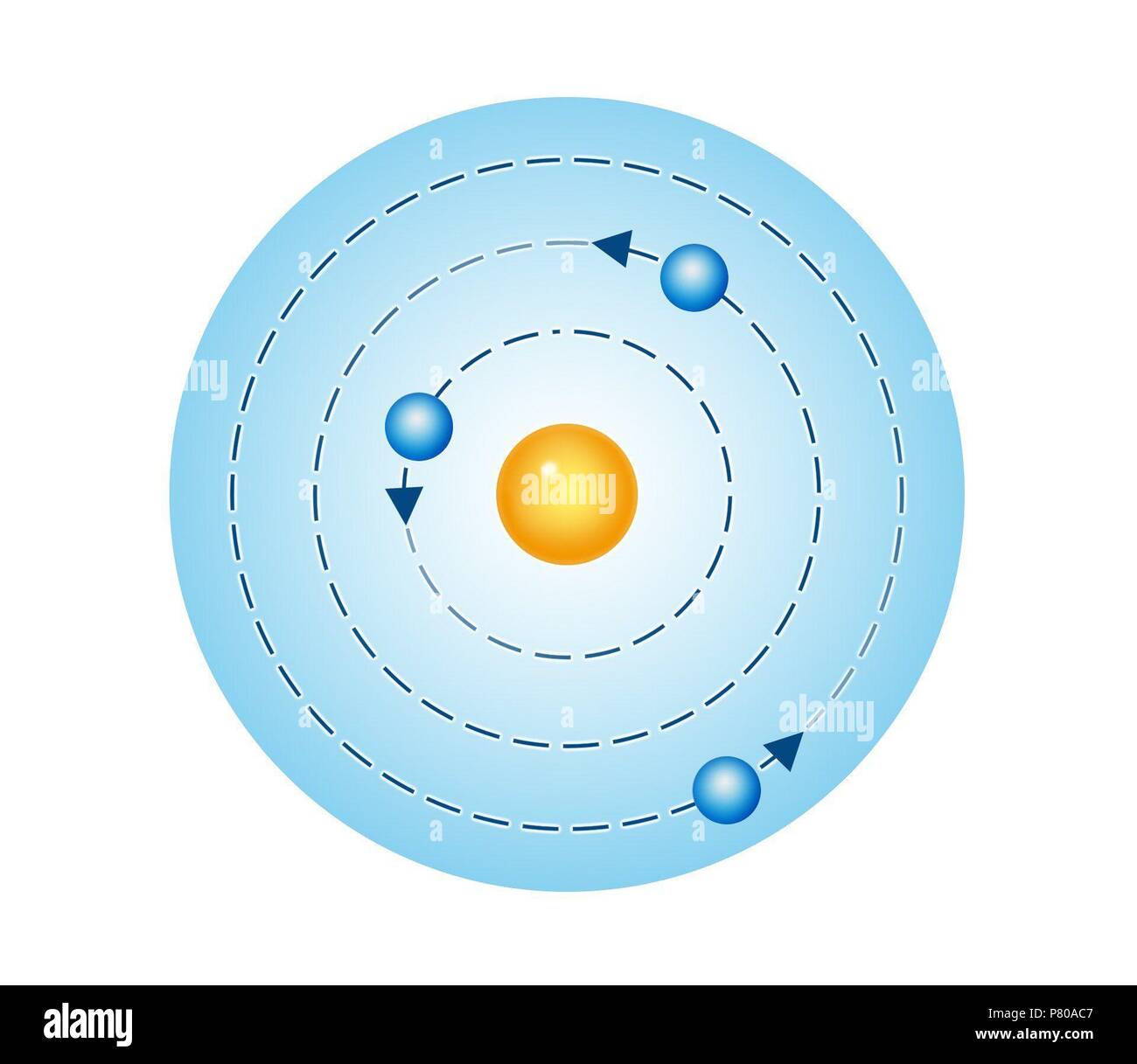
Classically, an electron continuously emitting energy should spiral into the nucleus. 
Bohr took this a step further, hypothesizing that the energy E is the difference in the atom’s energy when an electron moves from one orbit to another.įigure 3. However, he did recognize that the frequency of emitted radiation is determined by E = hf (actually, f = E/h). Interestingly, Bohr didn’t believe in photons when he developed the quantum view of the atom. Five years later Einstein proposed not only that material energy is quantized, but that light itself exists as quantum lumps, or “corpuscles,” later named photons. With the constant of proportionality h (Planck’s constant) we have the familiar E = hf. In 1900 Max Planck postulated that the energy of a radiated quantum of energy is proportional to the frequency of radiation: E ~ f. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
